To measure the density of any liquid substance, there is an instrument called a hydrometer. This density fluctuates (to a minor degree) when the temperature drops or rises than 4° C. At this point, the density of water is 1g/cm 3. Pure water reaches its maximum density at around 4☌. Why is the Density of Water Maximum at 4 Degree Celsius?

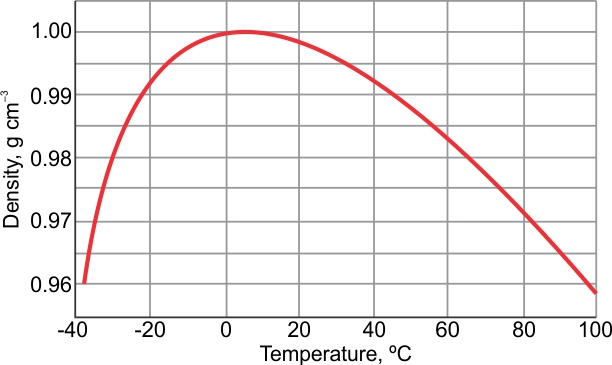
The maximum density of water is achieved when it is at a temperature of 4° C. When it comes to liquid water, the minimum density is in equilibrium with steam at 100° C. When water is in its vapour stage (steam form), it is at its minimum density. What is the Maximum and Minimum Density of Water? Only when the temperatures are on the extreme ends of the spectrum, for instance, if it is boiling or freezing, then only the change in density is noticeable enough. Therefore, this is the value that is mostly used in almost all types of density calculations. The range of temperature that is there in an indoor science lab (10° C – 21° C) mostly accounts for the density of the water to stay in and around 1 g/cm3. To help you understand how water changes its density in different temperatures, we have brought together the following chart. What is the Density of Water at Different Temperatures? This type of negative thermal expansion is associated with orientation-dependency, strong, interactions, or intermolecular forces, and it is observed in the form of molten silica. When water is further cooled, it becomes less dense and tends to expand. Liquid water becomes highly dense when it is cooled from room temperature. Instead of monotonic, it is unimodal by nature. The relation between the temperature of the water and its density is non-linear. Water has a higher density while it is in its liquid form than when it is in its solid form. This is why until and unless you are required to know the precise calculations or you are in a situation where a scientific experiment is going on that involves extreme pressure or temperature, you can keep using 1 g/cm 3 as the standard density of water. The variations in the density of water with changing temperatures are very subtle. The density of water is not absolute by nature since it keeps on changing with temperature. Why is the Density of Water Different at Different Temperatures? However, it should be noted that the exact density of water depends on the temperature of the area and the air pressure. Density is mass divided by volume, and since water is used as the basis for establishing the metric unit of mass, therefore one cubic centimeter (1 cm 3 ) of water weighs one gram (1g). In most given circumstances, the density of water is 1. The density of water can be defined as the weight of the water per its unit volume, which depends on the temperature of the water.
In other words, the density of a particular substance is basically the relationship between its mass and the volume it takes up. With the knowledge of the density of a particular substance, you can tell how tightly that substance is packed together. What is meant by the Density of Water?Īs we have stated above, the density of any substance refers to its mass per unit volume. But to take a closer look at understanding the density of water, we are going to uncover all related sub-topics under this category. At a simple glance, density refers to the mass per unit of a given substance. In today’s topic, we are going to try and understand one of its essential aspects – its density. Water continually moves through a cycle of evaporation, transpiration, condensation, precipitation, and runoff. This transparent, inorganic, odourless, and almost colourless chemical substance serves as the main constituent of the Earth’s hydrosphere. It is also present in the form of groundwater, glaciers, ice caps, clouds, vapours, and precipitation.įrom fishing to agriculture, water plays a pivotal role in every sphere of our economy and daily activities. It constitutes 71% of the Earth’s surface in the forms of oceans, seas, rivers, lakes, ponds, streams, and canals, among others. Cells, which are the basic structure of any life forms wouldn’t exist without water. Water is essential for all kinds of life forms on our planet, even though it is not known for providing any organic nutrients or calories.
